Imagine walking into a professional kitchen. The air is a chaotic mix of intense heat, acidic lemon juice, sharp knives, and constant moisture. In this environment, most materials would fall apart within weeks. Pure iron would flake away into orange dust, and copper would turn a dull, sickly green. Yet, the vast stretches of silvery counters and heavy pans stay gleaming, even after being pelted with salt and scrubbed with rough pads. This isn't just because chefs are good at cleaning; it's because the metal itself is performing a silent, microscopic act of healing every second of the day.
This metallurgical "magic" is so common that we have largely forgotten how revolutionary it is. Before the early 20th century, if you scratched a metal tool, that mark was a permanent invitation for the world to reclaim the object through rust. Today, you can take a steak knife, gouge a deep line into its surface, and within milliseconds, the metal essentially grows a new skin to seal the wound. To understand how stainless steel pulls this off, we have to look past the shiny surface and into a world of molecular shapes, where oxygen is both the enemy and the ultimate doctor.
The Chemistry of Crumbling Foundations
To appreciate why stainless steel is a hero, we first have to understand the fatal flaw of "normal" iron. Iron is an extroverted element; it desperately wants to bond with oxygen molecules in the air. When they meet, they form iron oxide, which we know as rust. The problem isn't just the change in color; it's the weakness of the new material. Iron oxide molecules are much larger and bulkier than the original iron atoms. As they form, they expand and push away from each other, creating a flaky, porous texture that looks like a pile of dried leaves.
This porous nature is the downfall of iron. Because rust is loose and full of tiny holes, it allows more oxygen and moisture to tunnel deeper into the metal, reaching the fresh iron underneath. It is a slow, relentless chain reaction of decay that moves from the surface to the core until the object loses its strength and snaps. Scientists call this an "active" corrosion process because the rust itself helps more rust to form. There is no biological scab or protective scar; there is only a growing, hungry rot.
The Chromium Sentinel and the Passive Layer
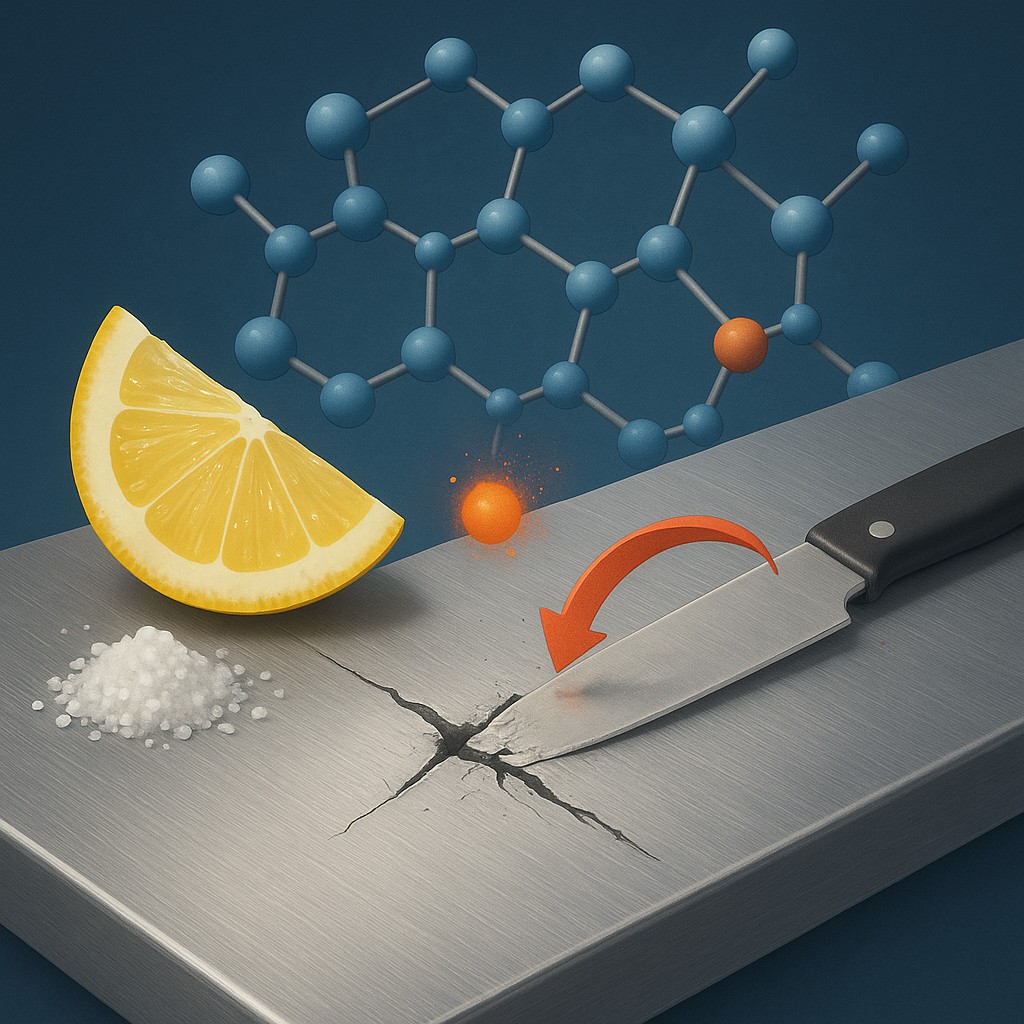
Stainless steel solves this problem with a secret ingredient: chromium. For steel to be called "stainless," it usually needs to contain at least 10 or 11 percent chromium. This specific ratio changes how the atoms interact. Chromium is even more eager to react with oxygen than iron is, but it does so in a much more organized way. When the chromium atoms on the surface hit the air, they bond together to form a microscopic film called chromium oxide.
Unlike loose and crumbly iron oxide, chromium oxide is incredibly tight. Imagine packing a suitcase: iron oxide is like throwing tangled hangers into the bag, leaving huge gaps and preventing the lid from closing. Chromium oxide is like vacuum-sealing your clothes into a flat, solid sheet. This layer, often called the "passive film," is only a few atoms thick, making it completely invisible. It doesn't dull the shine of the metal, yet it is so dense that oxygen molecules simply cannot squeeze through. It acts as a molecular shield, locking the iron safely behind a wall of chromium.
The Instant Miracle of Self-Repair
The most incredible part of this technology is how it reacts to damage. If you scratch a stainless steel fork against a plate, you are ripping through that protective chromium oxide skin. For a split second, the vulnerable iron and nickel atoms underneath are exposed to the air. In a regular piece of steel, this would be the beginning of the end. However, in stainless steel, the chromium atoms sitting just below the surface are suddenly exposed to oxygen for the first time.
Within milliseconds, these new chromium atoms react with the air and knit together a brand-new layer of chromium oxide. The "wound" is sealed before the iron even realizes it was exposed. This process happens automatically, without any help from us, and it can happen over and over again for decades. This is why stainless steel doesn't need to be painted like a bridge or a car frame. The protection isn't a coat of paint applied in a factory; it is a living part of the metal's chemical identity.
Environmental Weaknesses and Shield Failure
While stainless steel seems invincible, every hero has a weakness. For this metal, the enemy is often chlorine, which is found in seawater and industrial bleaches. Chloride ions are incredibly small and aggressive. They are the molecular equivalent of a specialized drill bit. In high concentrations, these ions can find tiny flaws in the chromium oxide skin and tunnel through faster than the chromium can repair the damage.
When this happens, the metal suffers from "pitting." Instead of a broad layer of rust, the damage shows up as tiny, deep holes that look like pinpricks but can travel deep into the structure. This is why you rarely see standard "304" grade stainless steel used on offshore oil rigs or boats. For those environments, engineers add more ingredients, like molybdenum, to the mix. These extra elements act like steel reinforcement for the chromium oxide layer, making it even tougher against chemical drills.
Comparing Common Metal Defenses
| Material |
Primary Defense |
Resilience to Scratches |
Long-term Maintenance |
| Carbon Steel |
Paint or Oil |
Very Low (Rust spreads from the cut) |
High (Needs frequent repainting) |
| Galvanized Steel |
Zinc Coating |
Moderate (Zinc erodes to save the steel) |
Medium (Protection eventually wears out) |
| Stainless Steel |
Chromium Oxide Film |
High (Self-heals in milliseconds) |
Low (Needs oxygen and cleaning) |
| Aluminum |
Aluminum Oxide Layer |
High (Similar to stainless steel) |
Low (Sensitive to high acidity) |
The Irony of Oxygen and the Need to Breathe
One of the most surprising facts about stainless steel is that it actually needs "fresh air" to stay healthy. Because the self-healing process relies on chromium reacting with oxygen, the metal can start to rust if you deprive it of air. This often happens in tight gaps or under rubber gaskets where water can seep in but air cannot. In these stagnant, oxygen-starved spots, the chromium cannot regrow its protective skin after a scratch. The metal then becomes "active" and begins to rot just like regular iron.
This leads to a surprising rule for taking care of stainless steel: it needs to stay clean and open to the air. When dirt, grease, or food sit on the surface for a long time, they create a mask that blocks oxygen. Professional chefs are taught to dry their stainless pans thoroughly and avoid leaving them to soak in water for days. By keeping the surface clear, you ensure the chromium "guards" always have the oxygen they need to maintain their invisible armor.
Seeing the World Through a New Lens
Understanding the "passive layer" helps us see the world not as a collection of solid, unchanging objects, but as a series of constant chemical negotiations. Your kitchen sink is not just a hunk of metal; it is a high-speed construction site where atoms are constantly bonding, breaking, and rebuilding. Every time you wash a pot or drop a spoon, you are part of a microscopic dance that has been perfected by a century of science. It is a reminder that some of the most sophisticated technology in our homes isn't in our phones, but in the tools we use to cook dinner.
The next time you see the sun glinting off a polished handrail or a sleek appliance, take a moment to appreciate the invisible shield at work. We live in a world designed to tear things apart, yet we have found a way to bake the cure directly into the metal itself. This blend of iron, chromium, and nickel is a testament to human ingenuity, proving that by understanding the laws of the universe, we can take common elements and turn them into something that is, for all practical purposes, immortal.