Imagine walking into a hospital where the most powerful drugs in the pharmacy have simply stopped working. This is not the plot of a dystopian novel; it is a modern reality known as antimicrobial resistance. For decades, we treated bacteria like a single, massive enemy that could be flattened with a chemical steamroller. We poured antibiotics into our bodies and our livestock, only to find that the survivors of those chemical wars have taught their offspring how to build better armor. These "superbugs" cause infections that are increasingly impossible to cure with standard medicine, leaving doctors to search for an alternative that does not rely on the failing logic of chemical destruction.
The solution, it turns out, is not a smarter chemical, but a biological hunter that has been perfecting its craft for billions of years. Meet the bacteriophage, or "phage" for short. These are viruses that do not affect humans, plants, or animals. They have a single, obsessive purpose: to find and destroy specific bacteria. While we have spent the last eighty years trying to win an arms race against bacterial evolution in the lab, nature has already provided a library of billions of specialized assassins. We are currently witnessing a historic shift in public health as agencies scale up the use of these living medicines to treat patients for whom traditional antibiotics are now little more than sugar pills.
The Viral Assassin in the Microscopic Shadows
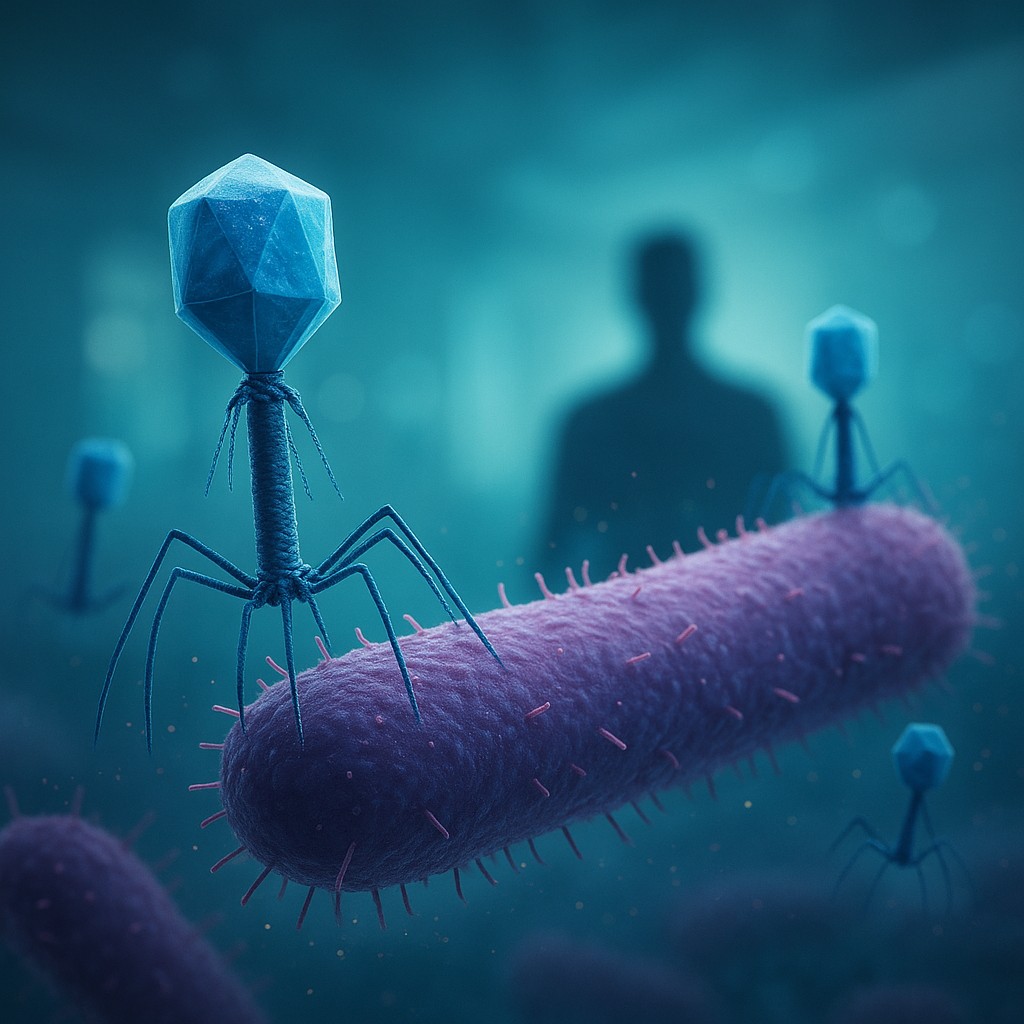
To understand why phages are the future of medicine, we first have to understand what they are and, more importantly, what they are not. A phage is essentially a genetic instruction manual wrapped in a protein shell, often looking remarkably like a tiny lunar lander. They are the most abundant life forms on the planet, found in every ecosystem from the deep ocean to the lining of your own throat. Unlike the viruses that cause the common cold or the flu, bacteriophages cannot infect human cells. Their biochemical "keys" only fit the "locks" found on the surfaces of specific bacteria.
When a phage finds a matching bacterium, the process is both elegant and brutal. It docks onto the surface of the cell and injects its own genetic material inside. Once this DNA or RNA enters the host, it effectively hijacks the bacterium’s machinery. Instead of the bacterium making more of itself, it is forced to become a factory for new phages. Hundreds of viral clones are assembled within the cell until the internal pressure becomes too great. At a precise moment, the phage produces an enzyme that dissolves the bacterial wall, causing the cell to physically burst - a process called lysis. This releases the new generation of phages into the surrounding environment, where they immediately begin hunting for more prey.
This mechanism represents a fundamental shift in how we think about "dosage." With a standard antibiotic like penicillin, the concentration of the drug in your system begins to drop the moment you take it, as your kidneys and liver work to filter it out. With phage therapy, the medicine is self-amplifying. As long as harmful bacteria are present, the phages will continue to replicate and increase their numbers. Once the target infection is cleared and there are no more hosts to infect, the phages have nothing left to do. They are eventually cleared from the body by the immune system or passed as waste, leaving the patient healed and the beneficial microbes intact.
The Scalpel Versus the Sledgehammer
The most significant advantage of phage therapy is its extreme precision. To appreciate this, we must consider the human microbiome - the trillions of microbes that live in our gut, on our skin, and in our lungs. Most of these bacteria are helpful or, at the very least, harmless. Traditional antibiotics are like carpet bombing from a high altitude; they kill the pathogen causing your pneumonia, but they also wipe out the "good" bacteria in your digestive tract. This ecological damage often leads to secondary issues like yeast infections or chronic stomach problems.
Phages, by contrast, function like a surgical strike team. Because a specific phage usually only targets a single strain or species of bacteria, it can hunt down the Salmonella or Staphylococcus causing an illness while leaving the rest of your internal ecosystem untouched. This personalized approach is why public health agencies are so invested in scaling this technology. However, this strength is also a logistical challenge. You cannot simply walk into a pharmacy and buy a "general phage" for a sore throat. Doctors must first grow a culture of the infection to find out exactly which bacterial strain is present and then "match-make" it with a phage from a vast library known to kill that specific strain.
| Feature |
Traditional Antibiotics |
Phage Therapy |
| Origin |
Chemical compounds (often fungal/synthetic) |
Naturally occurring viruses |
| Precision |
Broad-spectrum (kills many types) |
High-precision (targets specific strains) |
| Dosing |
Breaks down over time; requires repeat doses |
Self-replicating; increases while prey is present |
| Impact on Gut Health |
High collateral damage (kills good bacteria) |
Minimal to no impact on beneficial flora |
| Resistance |
Common; bacteria evolve chemical defenses |
Possible, but phages also evolve to counter them |
| Development |
Long, expensive chemical synthesis |
Rapid isolation from environmental sources |
Navigating Regulatory and Biological Hurdles
If phages are so effective, why haven't they replaced antibiotics already? The answer lies in the complexity of regulating a "living" drug. Our current medical infrastructure is built around the idea of stability. When health agencies approve a drug, they expect it to be a chemically identical molecule every time it is manufactured. Phages, as biological entities, are inherently variable. Furthermore, because phage therapy often needs to be customized for an individual patient, the traditional clinical trial model, which tests the same substance on thousands of people, is difficult to apply.
There is also the matter of the bacterial counter-response. Just as bacteria evolved to resist chemicals, they can also evolve to resist phages. They might change the proteins on their surface so the "lunar lander" can no longer dock, or they might develop internal immune systems, like CRISPR, to chop up the viral DNA. However, the beauty of phages is that they are also evolving. In a lab, scientists can "train" phages to overcome bacterial resistance in a matter of days. This creates a dynamic, evolutionary arms race where the medicine can actually outsmart the disease in real time.
Public health agencies are currently working on creating standardized "phage banks." Instead of starting from scratch for every patient, doctors could access a curated collection of phages that have already been screened for safety and purity. Recent initiatives in the UK, Belgium, and the United States are focused on streamlining the matching process. By using AI and high-speed screening, researchers can now compare the genetic signature of a patient's infection against thousands of candidate phages in a matter of hours, rather than weeks. This infrastructure is essential for moving phage therapy from a last-resort experimental treatment to a primary medical tool.
Search for Phages in the Wild
One of the most fascinating aspects of phage therapy is where the medicine comes from. Because phages live wherever bacteria thrive, the best place to find a phage that kills a specific human pathogen is often in places we usually avoid. To find phages that kill gut bacteria, researchers might look in raw sewage. To find phages that kill skin infections, they might sample soil or pond water. This gives the field a gritty, "prospecting" feel that is unlike the pristine, sterile environment of traditional pharmaceutical chemistry.
There are even "citizen science" programs where ordinary people collect water samples from their local environment to help build global phage libraries. This crowdsourced approach recognizes that the biodiversity of the microbial world is our greatest asset in the fight against superbugs. Once a promising phage is found, it is purified in a lab to ensure no bacterial toxins remain, and its genome is sequenced to make sure it doesn't carry any harmful genes. Only then is it ready to be used as a treatment.
It is helpful to think of this not as a replacement for antibiotics, but as a much-needed diversification of our medical options. In some cases, phages and antibiotics actually work better together. There is a phenomenon where a bacterium, in the process of evolving resistance to a phage, accidentally makes itself more vulnerable to an antibiotic it used to ignore. This "evolutionary trade-off" allows doctors to use a combination therapy to corner the bacteria, leaving them with no way to survive.
A New Era of Precision Medicine
We are moving away from the era of "one size fits all" medicine. The rise of phage therapy signals a deeper understanding of our place within a complex biological web. We are no longer just trying to sterilize our bodies; we are learning to manage our internal ecosystems with the help of specialized allies. By harnessing the natural rivalry between viruses and bacteria, we are turning an ancient biological war into a modern medical miracle.
As we scale this technology, the focus remains on accessibility and speed. The goal is a world where a patient with a drug-resistant infection doesn't have to wait for a miracle. Instead, their doctor will identify the specific culprit, pull the matching "viral assassin" from a digital and physical library, and deploy a treatment that is as precise as it is powerful. It is a testament to human ingenuity that the answer to our most modern medical crisis was hiding in the nearest puddle or backyard all along.
The journey from "germ theory" to "phage therapy" reflects our growing maturity as a species. We have learned that we cannot always win through brute force and chemical dominance. Sometimes, the most effective way to solve a problem is to find the natural process already designed to handle it and give it a helping hand. As you go about your day, remember that you are carrying an invisible army within you. Soon, our doctors will be able to recruit that army to fight the battles that chemistry alone can no longer win. Stay curious about the microscopic world, for it is there that the future of our health is being rewritten, one viral docking at a time.